countData <- read.csv("GSE37704_featurecounts.csv", row.names=1)
colData <- read.csv("GSE37704_metadata.csv", row.names=1)Class 14: RNASeq Mini- Project
Background
The data for today’s mini-project comes from a knock-down study of an important HOX gene.
Data Import
head(countData) length SRR493366 SRR493367 SRR493368 SRR493369 SRR493370
ENSG00000186092 918 0 0 0 0 0
ENSG00000279928 718 0 0 0 0 0
ENSG00000279457 1982 23 28 29 29 28
ENSG00000278566 939 0 0 0 0 0
ENSG00000273547 939 0 0 0 0 0
ENSG00000187634 3214 124 123 205 207 212
SRR493371
ENSG00000186092 0
ENSG00000279928 0
ENSG00000279457 46
ENSG00000278566 0
ENSG00000273547 0
ENSG00000187634 258head(colData) condition
SRR493366 control_sirna
SRR493367 control_sirna
SRR493368 control_sirna
SRR493369 hoxa1_kd
SRR493370 hoxa1_kd
SRR493371 hoxa1_kdClean up (data tidying)
We need to remove the length column from our countData to make the columns match the rows in colData.
countData <- countData[,-1]Check match of colData and countData
rownames(colData) == colnames(countData)[1] TRUE TRUE TRUE TRUE TRUE TRUEhead(countData) SRR493366 SRR493367 SRR493368 SRR493369 SRR493370 SRR493371
ENSG00000186092 0 0 0 0 0 0
ENSG00000279928 0 0 0 0 0 0
ENSG00000279457 23 28 29 29 28 46
ENSG00000278566 0 0 0 0 0 0
ENSG00000273547 0 0 0 0 0 0
ENSG00000187634 124 123 205 207 212 258Remove zero count genes
to.keep <- rowSums(countData) > 0
countData <- countData[to.keep,]DESeq Analysis
library(DESeq2)Setting up the DESeq object
dds <- DESeqDataSetFromMatrix(countData = countData,
colData = colData,
design = ~condition)Warning in DESeqDataSet(se, design = design, ignoreRank): some variables in
design formula are characters, converting to factorsRunning DESeq
dds <- DESeq(dds)estimating size factorsestimating dispersionsgene-wise dispersion estimatesmean-dispersion relationshipfinal dispersion estimatesfitting model and testingddsclass: DESeqDataSet
dim: 15975 6
metadata(1): version
assays(4): counts mu H cooks
rownames(15975): ENSG00000279457 ENSG00000187634 ... ENSG00000276345
ENSG00000271254
rowData names(22): baseMean baseVar ... deviance maxCooks
colnames(6): SRR493366 SRR493367 ... SRR493370 SRR493371
colData names(2): condition sizeFactorGetting results
res = results(dds)
head(res)log2 fold change (MLE): condition hoxa1 kd vs control sirna
Wald test p-value: condition hoxa1 kd vs control sirna
DataFrame with 6 rows and 6 columns
baseMean log2FoldChange lfcSE stat pvalue
<numeric> <numeric> <numeric> <numeric> <numeric>
ENSG00000279457 29.9136 0.1792571 0.3248216 0.551863 5.81042e-01
ENSG00000187634 183.2296 0.4264571 0.1402658 3.040350 2.36304e-03
ENSG00000188976 1651.1881 -0.6927205 0.0548465 -12.630158 1.43990e-36
ENSG00000187961 209.6379 0.7297556 0.1318599 5.534326 3.12428e-08
ENSG00000187583 47.2551 0.0405765 0.2718928 0.149237 8.81366e-01
ENSG00000187642 11.9798 0.5428105 0.5215598 1.040744 2.97994e-01
padj
<numeric>
ENSG00000279457 6.86555e-01
ENSG00000187634 5.15718e-03
ENSG00000188976 1.76549e-35
ENSG00000187961 1.13413e-07
ENSG00000187583 9.19031e-01
ENSG00000187642 4.03379e-01Volcano plot
A plot of log2 fold change vs -log of Adjusted P-value
library(ggplot2)ggplot(res) +
aes(log2FoldChange, -log(padj)) +
geom_point()Warning: Removed 1237 rows containing missing values or values outside the scale range
(`geom_point()`).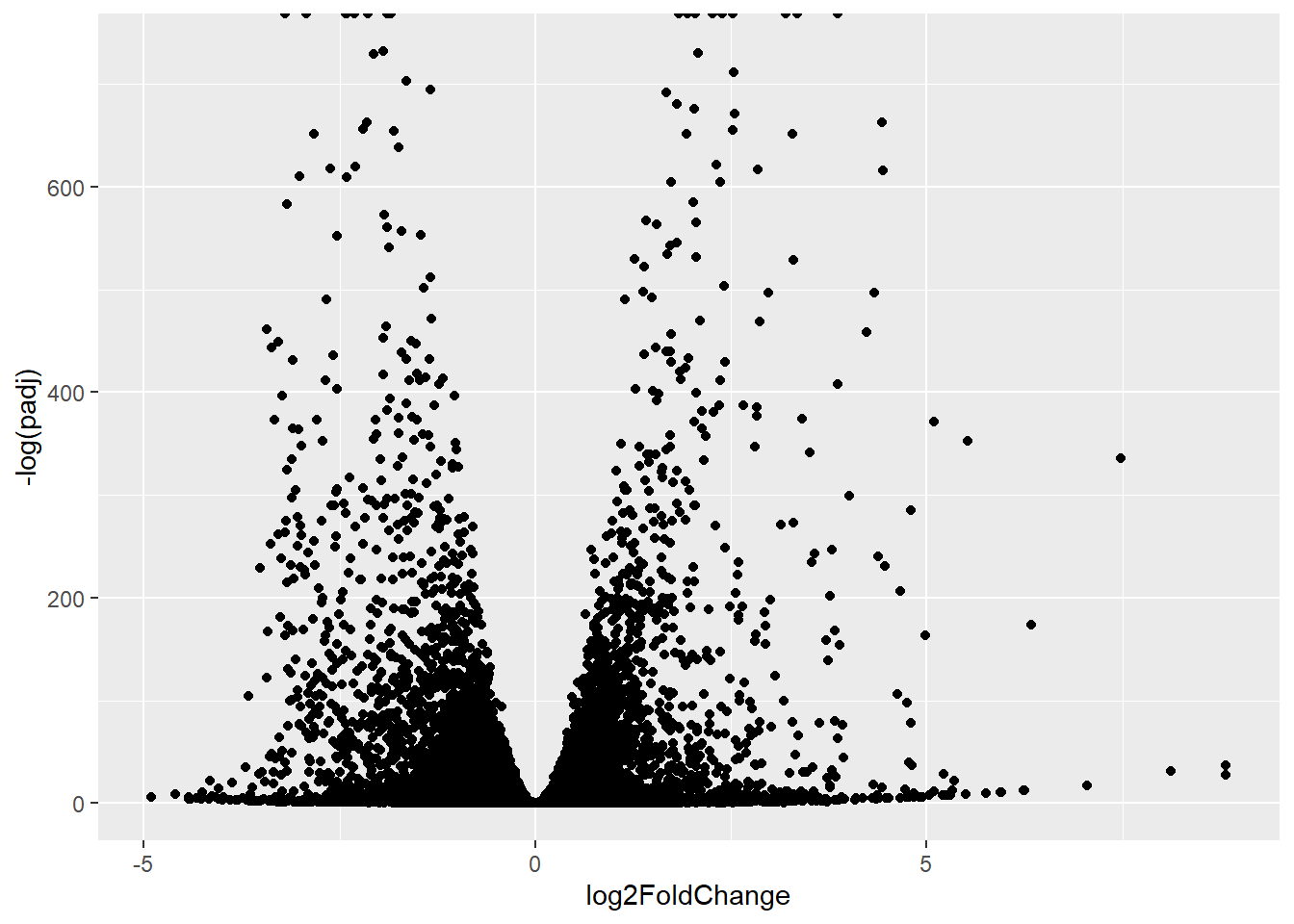
mycols <- rep("gray", nrow(res))
mycols[res$log2FoldChange > 2] <- "blue"
mycols[res$log2FoldChange < -2] <- "blue"
mycols[ res$padj >= 0.05 ] <- "gray"ggplot(res) +
aes(x= log2FoldChange, y= -log(padj)) +
geom_point(col = mycols) +
xlab("Log2(FoldChange)") +
ylab("-Log(P-value)") +
geom_vline(xintercept = c(-2,+2), col="red") +
geom_hline(yintercept = -log(0.05), col="red")Warning: Removed 1237 rows containing missing values or values outside the scale range
(`geom_point()`).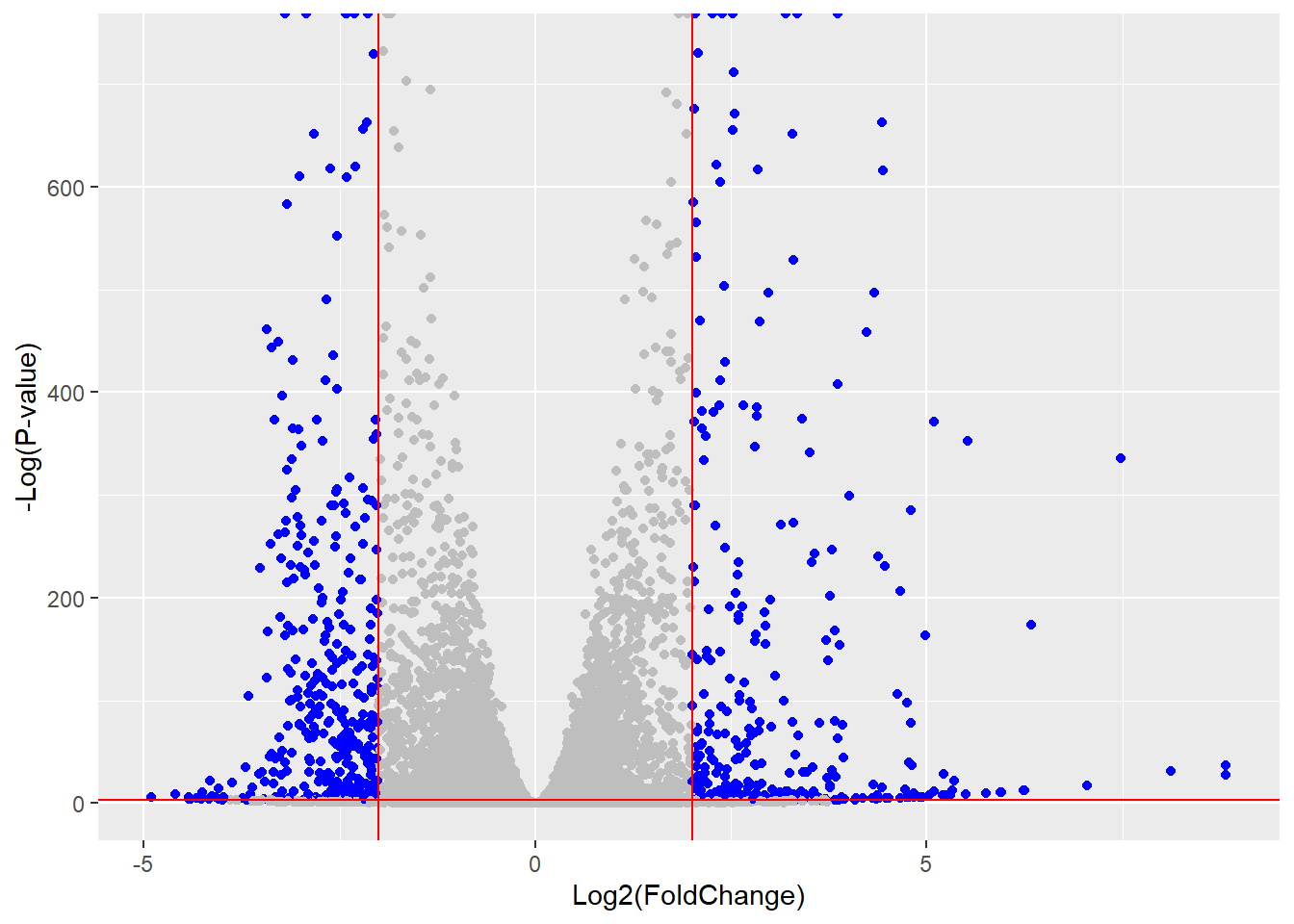
Add Annotation
library("AnnotationDbi")
library("org.Hs.eg.db")columns(org.Hs.eg.db) [1] "ACCNUM" "ALIAS" "ENSEMBL" "ENSEMBLPROT" "ENSEMBLTRANS"
[6] "ENTREZID" "ENZYME" "EVIDENCE" "EVIDENCEALL" "GENENAME"
[11] "GENETYPE" "GO" "GOALL" "IPI" "MAP"
[16] "OMIM" "ONTOLOGY" "ONTOLOGYALL" "PATH" "PFAM"
[21] "PMID" "PROSITE" "REFSEQ" "SYMBOL" "UCSCKG"
[26] "UNIPROT" res$symbol = mapIds(org.Hs.eg.db,
keys=row.names(res),
keytype="ENSEMBL",
column="SYMBOL",
multiVals="first")'select()' returned 1:many mapping between keys and columnsres$entrez = mapIds(org.Hs.eg.db,
keys=row.names(res),
keytype="ENSEMBL",
column="ENTREZID",
multiVals="first")'select()' returned 1:many mapping between keys and columnsres$name = mapIds(org.Hs.eg.db,
keys=row.names(res),
keytype="ENSEMBL",
column="GENENAME",
multiVals="first")'select()' returned 1:many mapping between keys and columnshead(res, 10)log2 fold change (MLE): condition hoxa1 kd vs control sirna
Wald test p-value: condition hoxa1 kd vs control sirna
DataFrame with 10 rows and 9 columns
baseMean log2FoldChange lfcSE stat pvalue
<numeric> <numeric> <numeric> <numeric> <numeric>
ENSG00000279457 29.913579 0.1792571 0.3248216 0.551863 5.81042e-01
ENSG00000187634 183.229650 0.4264571 0.1402658 3.040350 2.36304e-03
ENSG00000188976 1651.188076 -0.6927205 0.0548465 -12.630158 1.43990e-36
ENSG00000187961 209.637938 0.7297556 0.1318599 5.534326 3.12428e-08
ENSG00000187583 47.255123 0.0405765 0.2718928 0.149237 8.81366e-01
ENSG00000187642 11.979750 0.5428105 0.5215598 1.040744 2.97994e-01
ENSG00000188290 108.922128 2.0570638 0.1969053 10.446970 1.51282e-25
ENSG00000187608 350.716868 0.2573837 0.1027266 2.505522 1.22271e-02
ENSG00000188157 9128.439422 0.3899088 0.0467163 8.346304 7.04321e-17
ENSG00000237330 0.158192 0.7859552 4.0804729 0.192614 8.47261e-01
padj symbol entrez name
<numeric> <character> <character> <character>
ENSG00000279457 6.86555e-01 NA NA NA
ENSG00000187634 5.15718e-03 SAMD11 148398 sterile alpha motif ..
ENSG00000188976 1.76549e-35 NOC2L 26155 NOC2 like nucleolar ..
ENSG00000187961 1.13413e-07 KLHL17 339451 kelch like family me..
ENSG00000187583 9.19031e-01 PLEKHN1 84069 pleckstrin homology ..
ENSG00000187642 4.03379e-01 PERM1 84808 PPARGC1 and ESRR ind..
ENSG00000188290 1.30538e-24 HES4 57801 hes family bHLH tran..
ENSG00000187608 2.37452e-02 ISG15 9636 ISG15 ubiquitin like..
ENSG00000188157 4.21963e-16 AGRN 375790 agrin
ENSG00000237330 NA RNF223 401934 ring finger protein ..res = res[order(res$pvalue),]
write.csv(res, file="deseq_results.csv")Pathway Analysis
library(pathview)##############################################################################
Pathview is an open source software package distributed under GNU General
Public License version 3 (GPLv3). Details of GPLv3 is available at
http://www.gnu.org/licenses/gpl-3.0.html. Particullary, users are required to
formally cite the original Pathview paper (not just mention it) in publications
or products. For details, do citation("pathview") within R.
The pathview downloads and uses KEGG data. Non-academic uses may require a KEGG
license agreement (details at http://www.kegg.jp/kegg/legal.html).
##############################################################################library(gage)library(gageData)data(kegg.sets.hs)
data(sigmet.idx.hs)KEGG
# Focus on signaling and metabolic pathways only
kegg.sets.hs = kegg.sets.hs[sigmet.idx.hs]
# Examine the first 3 pathways
head(kegg.sets.hs, 3)$`hsa00232 Caffeine metabolism`
[1] "10" "1544" "1548" "1549" "1553" "7498" "9"
$`hsa00983 Drug metabolism - other enzymes`
[1] "10" "1066" "10720" "10941" "151531" "1548" "1549" "1551"
[9] "1553" "1576" "1577" "1806" "1807" "1890" "221223" "2990"
[17] "3251" "3614" "3615" "3704" "51733" "54490" "54575" "54576"
[25] "54577" "54578" "54579" "54600" "54657" "54658" "54659" "54963"
[33] "574537" "64816" "7083" "7084" "7172" "7363" "7364" "7365"
[41] "7366" "7367" "7371" "7372" "7378" "7498" "79799" "83549"
[49] "8824" "8833" "9" "978"
$`hsa00230 Purine metabolism`
[1] "100" "10201" "10606" "10621" "10622" "10623" "107" "10714"
[9] "108" "10846" "109" "111" "11128" "11164" "112" "113"
[17] "114" "115" "122481" "122622" "124583" "132" "158" "159"
[25] "1633" "171568" "1716" "196883" "203" "204" "205" "221823"
[33] "2272" "22978" "23649" "246721" "25885" "2618" "26289" "270"
[41] "271" "27115" "272" "2766" "2977" "2982" "2983" "2984"
[49] "2986" "2987" "29922" "3000" "30833" "30834" "318" "3251"
[57] "353" "3614" "3615" "3704" "377841" "471" "4830" "4831"
[65] "4832" "4833" "4860" "4881" "4882" "4907" "50484" "50940"
[73] "51082" "51251" "51292" "5136" "5137" "5138" "5139" "5140"
[81] "5141" "5142" "5143" "5144" "5145" "5146" "5147" "5148"
[89] "5149" "5150" "5151" "5152" "5153" "5158" "5167" "5169"
[97] "51728" "5198" "5236" "5313" "5315" "53343" "54107" "5422"
[105] "5424" "5425" "5426" "5427" "5430" "5431" "5432" "5433"
[113] "5434" "5435" "5436" "5437" "5438" "5439" "5440" "5441"
[121] "5471" "548644" "55276" "5557" "5558" "55703" "55811" "55821"
[129] "5631" "5634" "56655" "56953" "56985" "57804" "58497" "6240"
[137] "6241" "64425" "646625" "654364" "661" "7498" "8382" "84172"
[145] "84265" "84284" "84618" "8622" "8654" "87178" "8833" "9060"
[153] "9061" "93034" "953" "9533" "954" "955" "956" "957"
[161] "9583" "9615" foldchanges = res$log2FoldChange
names(foldchanges) = res$entrez
head(foldchanges) 1266 54855 1465 2034 2150 6659
-2.422719 3.201955 -2.313738 -1.888019 3.344508 2.392288 keggres = gage(foldchanges, gsets=kegg.sets.hs)attributes(keggres)$names
[1] "greater" "less" "stats" head(keggres$less) p.geomean stat.mean p.val
hsa04110 Cell cycle 8.995727e-06 -4.378644 8.995727e-06
hsa03030 DNA replication 9.424076e-05 -3.951803 9.424076e-05
hsa03013 RNA transport 1.375901e-03 -3.028500 1.375901e-03
hsa03440 Homologous recombination 3.066756e-03 -2.852899 3.066756e-03
hsa04114 Oocyte meiosis 3.784520e-03 -2.698128 3.784520e-03
hsa00010 Glycolysis / Gluconeogenesis 8.961413e-03 -2.405398 8.961413e-03
q.val set.size exp1
hsa04110 Cell cycle 0.001448312 121 8.995727e-06
hsa03030 DNA replication 0.007586381 36 9.424076e-05
hsa03013 RNA transport 0.073840037 144 1.375901e-03
hsa03440 Homologous recombination 0.121861535 28 3.066756e-03
hsa04114 Oocyte meiosis 0.121861535 102 3.784520e-03
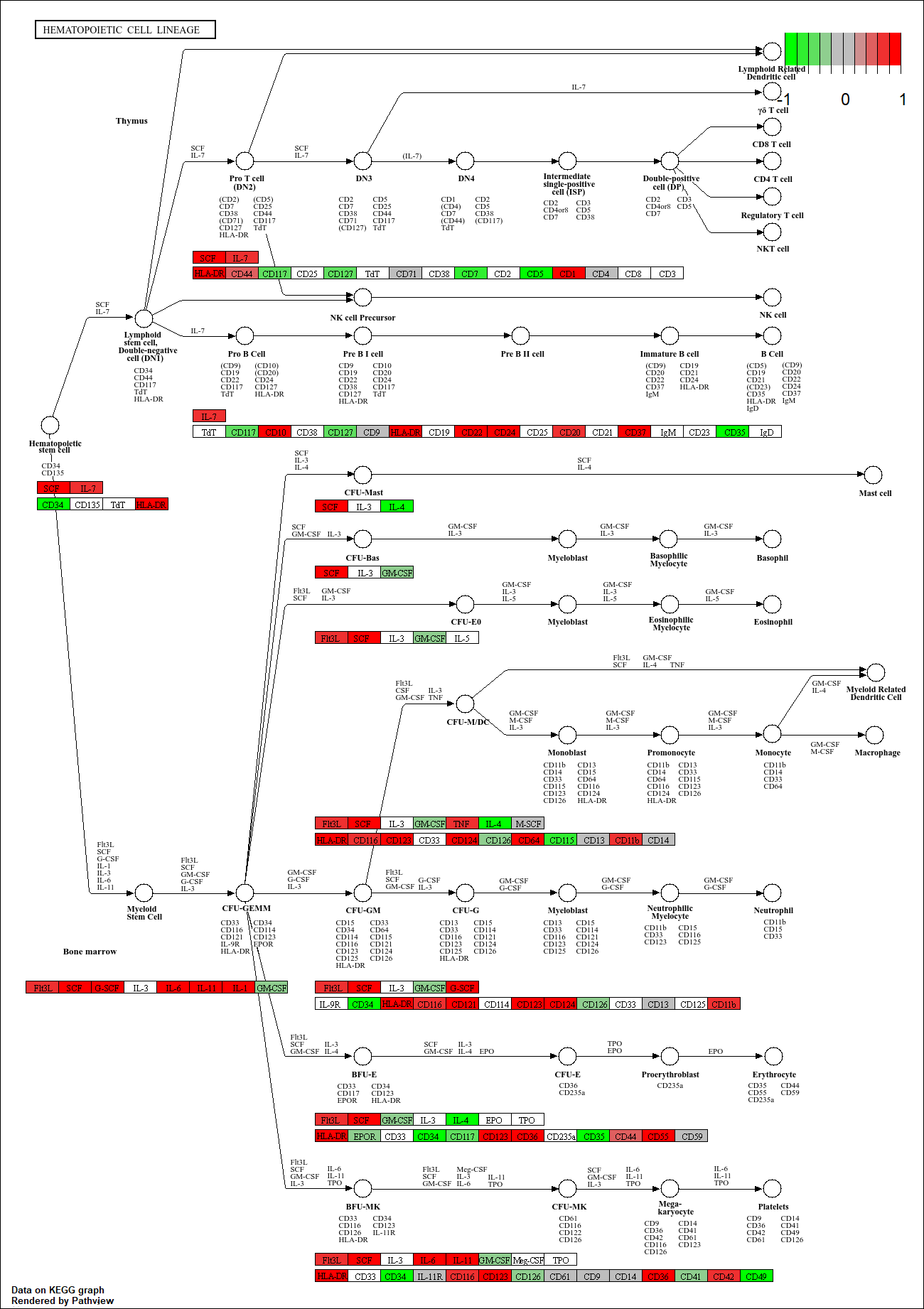
hsa00010 Glycolysis / Gluconeogenesis 0.212222694 53 8.961413e-03pathview(gene.data=foldchanges, pathway.id="hsa04110")'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04110.pathview.png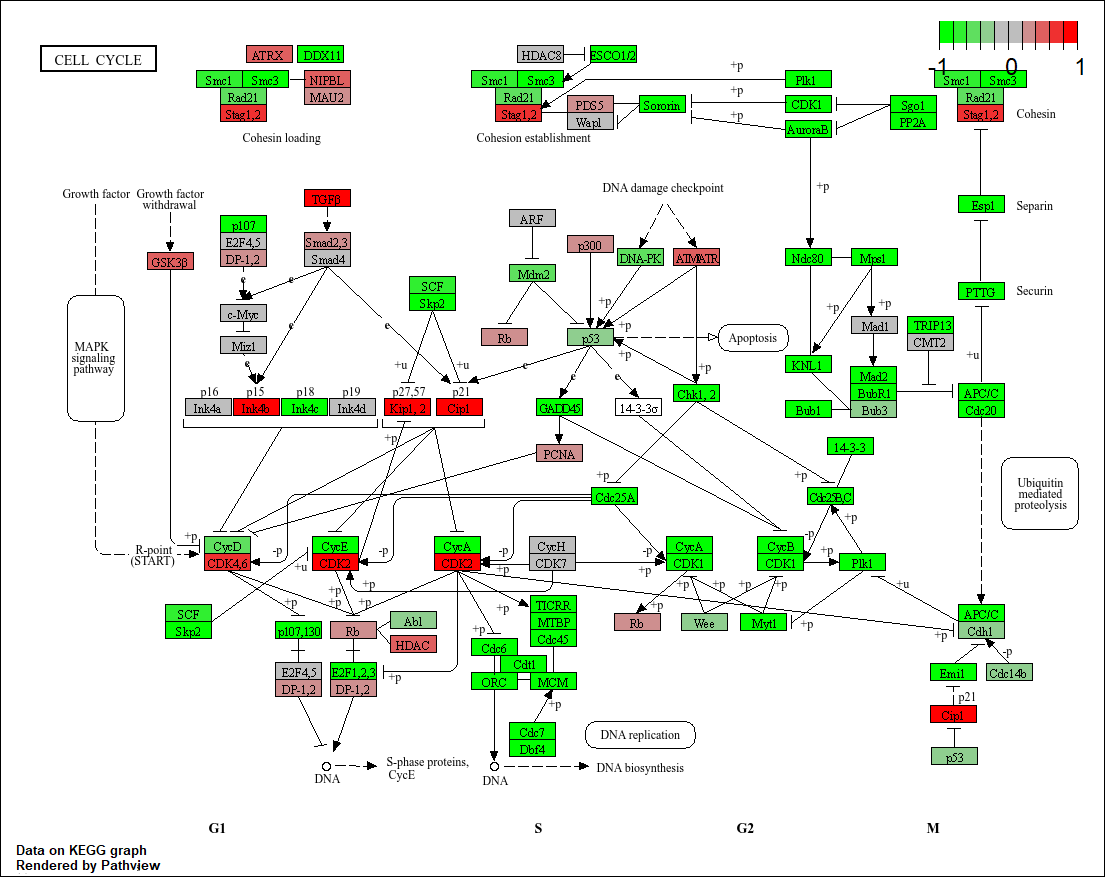
pathview(gene.data=foldchanges, pathway.id="hsa04110", kegg.native=FALSE)'select()' returned 1:1 mapping between keys and columnsWarning: reconcile groups sharing member nodes! [,1] [,2]
[1,] "9" "300"
[2,] "9" "306"Info: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04110.pathview.pdf## Focus on top 5 upregulated pathways here for demo purposes only
keggrespathways <- rownames(keggres$greater)[1:5]
# Extract the 8 character long IDs part of each string
keggresids = substr(keggrespathways, start=1, stop=8)
keggresids[1] "hsa04640" "hsa04630" "hsa00140" "hsa04142" "hsa04330"pathview(gene.data=foldchanges, pathway.id=keggresids, species="hsa")'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04640.pathview.png'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04630.pathview.png'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa00140.pathview.pngInfo: Downloading xml files for hsa04142, 1/1 pathways..Info: Downloading png files for hsa04142, 1/1 pathways..'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04142.pathview.pngInfo: Downloading xml files for hsa04330, 1/1 pathways..Info: Downloading png files for hsa04330, 1/1 pathways..'select()' returned 1:1 mapping between keys and columnsInfo: Working in directory C:/Users/patri/OneDrive/Desktop/Bimm143/class14Info: Writing image file hsa04330.pathview.png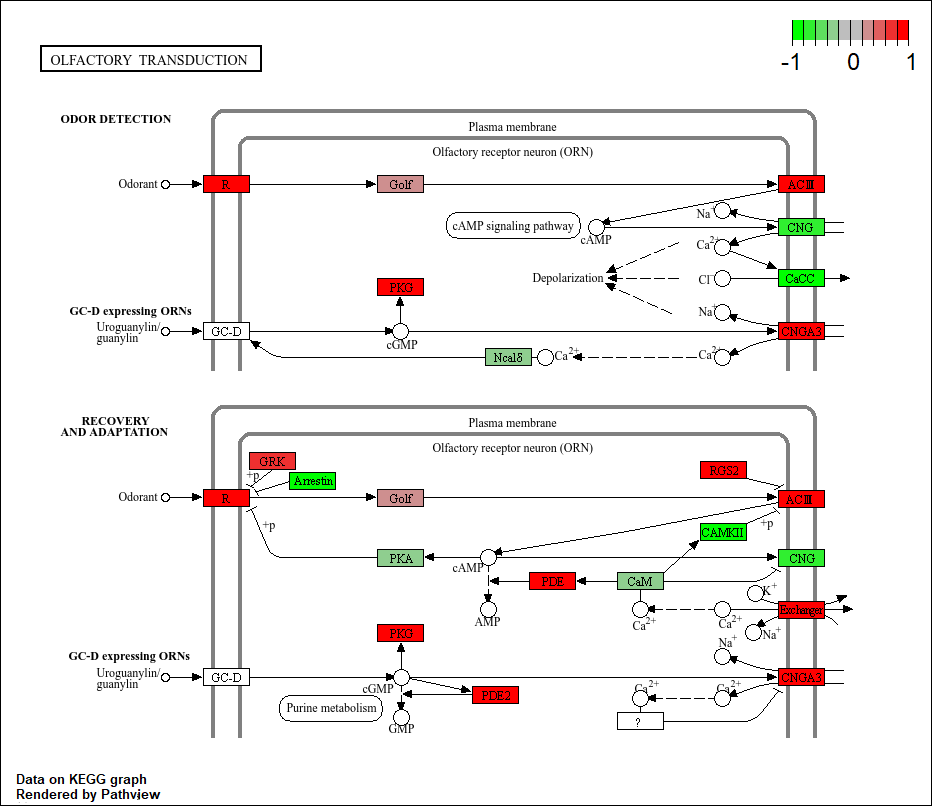
GO
data(go.sets.hs)
data(go.subs.hs)
# Focus on Biological Process subset of GO
gobpsets = go.sets.hs[go.subs.hs$BP]
gobpres = gage(foldchanges, gsets=gobpsets)
lapply(gobpres, head)$greater
p.geomean stat.mean p.val
GO:0007156 homophilic cell adhesion 8.519724e-05 3.824205 8.519724e-05
GO:0002009 morphogenesis of an epithelium 1.396681e-04 3.653886 1.396681e-04
GO:0048729 tissue morphogenesis 1.432451e-04 3.643242 1.432451e-04
GO:0007610 behavior 1.925222e-04 3.565432 1.925222e-04
GO:0060562 epithelial tube morphogenesis 5.932837e-04 3.261376 5.932837e-04
GO:0035295 tube development 5.953254e-04 3.253665 5.953254e-04
q.val set.size exp1
GO:0007156 homophilic cell adhesion 0.1951953 113 8.519724e-05
GO:0002009 morphogenesis of an epithelium 0.1951953 339 1.396681e-04
GO:0048729 tissue morphogenesis 0.1951953 424 1.432451e-04
GO:0007610 behavior 0.1967577 426 1.925222e-04
GO:0060562 epithelial tube morphogenesis 0.3565320 257 5.932837e-04
GO:0035295 tube development 0.3565320 391 5.953254e-04
$less
p.geomean stat.mean p.val
GO:0048285 organelle fission 1.536227e-15 -8.063910 1.536227e-15
GO:0000280 nuclear division 4.286961e-15 -7.939217 4.286961e-15
GO:0007067 mitosis 4.286961e-15 -7.939217 4.286961e-15
GO:0000087 M phase of mitotic cell cycle 1.169934e-14 -7.797496 1.169934e-14
GO:0007059 chromosome segregation 2.028624e-11 -6.878340 2.028624e-11
GO:0000236 mitotic prometaphase 1.729553e-10 -6.695966 1.729553e-10
q.val set.size exp1
GO:0048285 organelle fission 5.841698e-12 376 1.536227e-15
GO:0000280 nuclear division 5.841698e-12 352 4.286961e-15
GO:0007067 mitosis 5.841698e-12 352 4.286961e-15
GO:0000087 M phase of mitotic cell cycle 1.195672e-11 362 1.169934e-14
GO:0007059 chromosome segregation 1.658603e-08 142 2.028624e-11
GO:0000236 mitotic prometaphase 1.178402e-07 84 1.729553e-10
$stats
stat.mean exp1
GO:0007156 homophilic cell adhesion 3.824205 3.824205
GO:0002009 morphogenesis of an epithelium 3.653886 3.653886
GO:0048729 tissue morphogenesis 3.643242 3.643242
GO:0007610 behavior 3.565432 3.565432
GO:0060562 epithelial tube morphogenesis 3.261376 3.261376
GO:0035295 tube development 3.253665 3.253665Reactome
sig_genes <- res[res$padj <= 0.05 & !is.na(res$padj), "symbol"]
print(paste("Total number of significant genes:", length(sig_genes)))[1] "Total number of significant genes: 8147"write.table(sig_genes, file="significant_genes.txt", row.names=FALSE, col.names=FALSE, quote=FALSE).